Examples of Extrinsic Factors Affecting WHC of Processed Meats
- Physiological state of the meat: For many years before mechanical refrigeration was available, meat processors had to process meat the same day of slaughter. They soon learned that pre-rigor muscle was more functional than post-rigor meat, thus processing time became an important control factor. The reason for this was that the meat was less contracted, the water holding protein (myosin) was easier to extract because there were fewer actomyosin bonds between the contractile myofilaments, the proteins were less denatured, and the meat had a pH greater than normal. If pre-rigor processing is an option, it can significantly improve product WHC.
- Processing procedures: Various physical processes, such as mincing, chopping, mixing, vacuum tumbling, and massaging are necessary processes, but they disrupt the cellular structural integrity and open channels where water may be lost. The benefit of rapid chilling of carcasses was mentioned earlier, but maintaining consistently cold storage temperatures will decrease drip losses. If a product has to be frozen, rapid freezing and constant temperature during frozen storage also reduce drip. It should be clear that creative and controlled meat processing of fresh and processed meat products can pay dividends with improved yields.
- Aging of meat: For centuries meat has been aged to improve sensory properties. Only recently has research shown that proteolysis during aging was also related to WHC. Muscle has numerous protein linkages between cells and between muscle cells and the cell membrane and thin endomysial layer of connective tissue. If these small linkages do not undergo proteolysis early post-mortem, then WHC decreases. Additional work is needed to develop schemes of meat processing to accelerate degradation of these proteins to improve WHC. It appears that several key factors of WHC may also influence proteolysis of structural linkages.
- Processing Ingredients: The addition of salt and/or phosphates to meat has been done for many years to add flavor, serve as a preservative, and as enhancers of yield and bind of water. In general, "salts" of all compositions dissolving into the water and provide charged ions that can interact with the protein and water. For example "table salt" (sodium chloride, NaCl) will provide positive Na ions and negative Cl ions, the latter of which has a greater affect on increasing WHC because chloride is a more interactive ion with meat proteins than sodium.
Salt has the following effects on WHC:
- Salt will increase the total amount of charged ions that can interact with charges on the proteins.
- The isoelectric pH of the muscle proteins shift to a more acidic pH. Thus, meat with a normal pH will be farther from the isoelectric pH which increases WHC.
- Salt slightly increases muscle pH.
- Salt increases muscle filament spacing by providing ions that are attracted to charges on the protein surfaces. This widens the structural spacing between filaments, and allows the filaments to swell, providing more room for water molecules to flow into the spaces.
- Monovalent sodium ions replace some tightly bound divalent positive ions, thus increasing the ability of the filaments to swell.
- Salt frees more reactive groups on protein to hold to water.
Phosphates are commonly used in processed meats to help bind water. They do this by several mechanisms:
- Alkaline phosphates will elevate the pH 0.2 to 0.5 pH units higher than normal.
- At the higher pH, the negative phosphate ions (usually a negative 2 or 3 valence charge) contribute to higher charge on the muscle proteins.
- The negative phosphate ions can counter some of the divalent positive ions in meat.
- Some phosphates may help break some of the contraction cross bridges which increases WHC.
- Salt and phosphates act synergistically to increase WHC more the either salt or phosphate alone.
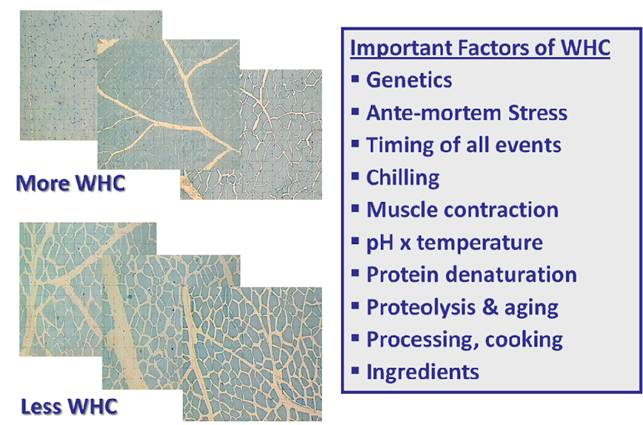
Figure III-3. Summary of Factors Affecting WHC. By now you should be able to integrate and evaluate the muscle structural and chemical factors listed in this Fig. III-3.