Salmonella in pig farms
Most of the pork in EU is coming from pigs raised in industrialised farms, following a number of systems and capitalising on the latest achievements of animal husbandry and farm management, in order to achieve maximum production, less health issues and potentially better animal welfare. In the next sections, some basic features of this type of farming will be provided to help the reader understand better the risks of such type of farming, as well as to understand which and why are the best control strategies under these farming conditions.
Industrialised pig farms
Nowadays, for a number of different reasons and for breed standardisation, there is general 3-tier system that is operating in pig farming (Figure 3). Nucleus herds are at the top level and they are responsible for producing pure breed animals, which are sold to breeding farms. Breeding farms are used to breed animals of the same of different breeds, and the offspring is sold to the fattening herds. The purpose of the fattening herds is to produce pigs that will be introduced into the pork production chain. The main risk regarding food safety arises from the fattening herds; however the distribution system of the pigs may be also an important factor in introduction of Salmonella into the fattening farms.
Figure 3. Distribution of pigs among different types of farms. From top to bottom, nucleus herds, breeding herds, and fattening herds.
The main stages of the life of a newborn piglet are the stage with the sow, the weaning phase and the fattening phase. These can be further subdivided into smaller stages, but this is out of scope of the material. For each stage there is a spatial change, meaning that piglets are moved from one room to another, more suitable for their needs depending on the stage of life they are. The fattening farms may or may not have all the stages of fattening. Indeed there are farms which buy piglets at the weaning or the fattening phase from other finishing farms.
Except of the stage where the piglets are still with the sow, the rooms in the next stages have an infrastructure similar to the one presented in Figure 4. Each house is divided in a number of rooms and each of the rooms is sub-divided in a number of pens. The contact of pigs living in different rooms is not feasible, while the contact among pigs living in the same room is limited.
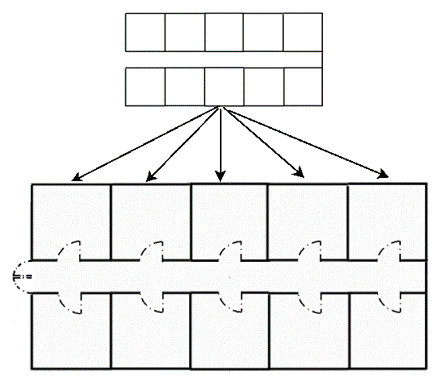
Figure 4. Infrastructure of a house in a common modern industrialized pig farm with intensive management. In the lower figure the infrastructure of a house is presented, while in the upper figure the typical infrastructure of a compartment/room of the house divided in pens is shown
Under an All-In-All-Out (AIAO) farming strategy, all pigs in a room are approximately of the same age and they enter and leave the room generally on the same day. In this way, there is the advantage of being able to clean and disinfect the room between the change of the batches of pigs increasing the probability of discontinuing whatever infection there is in that room.
Sources of Salmonella
One of the major sources of Salmonella in fattening farms as already pointed out above is the distribution of pigs and the pigs arriving from other farms, either from higher level or from the same level with the purpose of fattening.
A second important route of introduction of Salmonella in a room is the mechanical transfer. This can be attributed either to the farm staff, which moves from room to room or vectors such as vermin, given that the vector range for salmonellae is considered generally broad.
External sources of the pathogen may be transient vectors such as rodents, visitors, feed and animal vehicles or contaminated feed or water (Griffith et al., 2006). Finally, Salmonella may survive also in the external environment of the farm for a long period of time and can be continuously re-introduced into the fattening rooms.
For an AIAO production system, the survival of Salmonella after a room is cleaned could be also an important factor. The introduction of a new batch of pigs to a highly contaminated environment could lead to infection of this batch.
Finally, the transfer of pigs from room to room, because of delayed growth or belligerence of some pigs, reduces the barriers and opens more gates for Salmonella at the semi-closed system of the pen/room.
Pathogenesis and symptoms
As already referred above Salmonella is a pathogen of the intestinal tract and the main symptoms may arise from the gastro-intestinal system. S. Cholerasuis gives more overt symptoms and may lead to reduction of growth rate, increased mortality, etc. However, S. Typhimurium leads to a silent infection, which usually is not understood in the farm.
In short the pathogenesis has as follows: a pig can get infected after ingesting a large number of microorganisms. This intestinal infection in turn leads to the reproduction of Salmonella and shedding of the pathogen in high numbers for a period time. Salmonella also can be found at the local lymph nodes and survive there for a longer period of time. This infection triggers the immune system of the body which starts creating antibodies, which depending on the serotype may last for up to 110 days (Nielsen et al., 1995). More details on the pathogenesis can be found at Boyen et al. (2008, 2009) where it is supported that "Salmonella Typhimurium infection is dose-dependent".
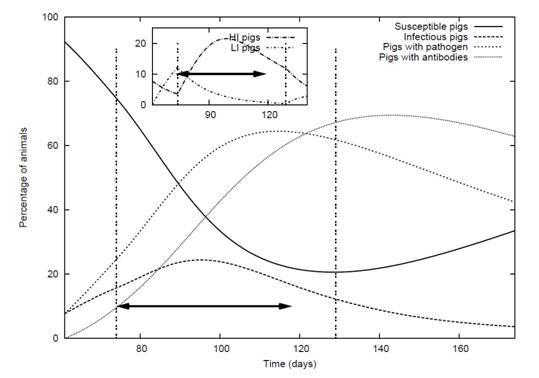
Figure 5. An example of a room of pigs which had 10% infected in the 60th day of fattening. This figure is a forecast using simulation and mathematical modelling and agreed well with experimental results (Soumpasis & Butler, 2011)
At the population level, not all pigs are getting infected at the same time. Thus, in a room where pigs are infected, it is expected that the animals would be in one of the different stages of infection: Susceptible (pigs with no infection), Infected (shedder pigs), Carriers (pigs with the pathogen) and Recovered (Pigs with antibodies). An example of such a situation is presented in Figure 5. Infected pigs can be further differentiated to pigs that have a stronger infection which can present also symptoms (HI pigs) and pigs with a lighter infection and no over symptoms (LI pigs in subfigure in figure 5). The effect of the infection depends more on the levels of the pathogen in the environment following the above mentioned statement that the infection with Salmonella is dose dependent. Although, this dose-dependency may be a continuous relationship, the categorisation of infection in two levels helps the reader in better understanding this concept and has been proven successful in terms of modelling the dynamics of the infection by providing good predicting results with small losses in accuracy.
Testing of pigs under each country's national monitoring programs leads to discovery of farms that have animals that are sero-positive to Salmonella. The testing for the national monitoring programs gives a picture of the last day of fattening. In this particular example used above (figure 5), 60% of the pigs are sero-positive at the last day of fattening. This value is reflecting the mean estimation of the prevalence of the pig with antibodies. In reality a number of different outcomes may exist, given the stochastic nature of the event (process noise). Using stochastic stimulation for this particular example, it was estimated that the possible numbers of pigs with antibodies at the time slaughter could range from 48% to 75% (Soumpasis & Butler, 2009).
Given that only a small number of pigs out of the total number that constitutes a batch are tested (usually around 10%), the results of the serological tests could show even more variation around the real values (observational noise). For this reason, the test schemes include usually a moving average to account indirectly for these uncertainties. Simply speaking, a single high value in three consecutive tests may be either a random effect or the beginning of a problem. The next tests will define the real origin of this value. Depending on the characterisation of the farm, further actions may have to be taken depending on the country and the national program that it is followed.
It is worth mentioning that even in heavily infected farms, clinical symptoms usually lack. However, it is still clear that an infection is present given the reproduction and transmission of Salmonella in the livestock. From analytical and descriptive studies (Leontides et al., 2003; Lo Fo Wong et al., 2004), it was observed that most of the highly contaminated farms, they had breaches of the bio-security measures.
Diagnostic Methods
Generally diagnosis of Salmonella is quite difficult, especially at the level of the individual animal. First, because the symptoms are quite general or the infection is silent. Second the isolation of the micro-organism in the environment is quite difficult. Finally, even if there is isolation of the pathogen from the environment, it does not necessary mean that there is an infection, given the Salmonella is widely spread and has quite high survival rates.
The techniques of collecting samples from the farm are mainly focused on the isolation of the pathogen from faecal samples from the animal or from the environment. Rectal swabs can ensure that the samples are coming from one individual, however they have very low sensitivity given the small sample size (in grams) and that the pathogen may not be shedding continuously (Arnold et al., 2005). An improvement of this method is the collection of "fresh" faeces, which allows for larger sample sizes (van der Wolf et al., 1999).
In order to avoid the disadvantages of the above methods, researchers often prefer environmental or "pooled" faecal samples (Arnold et al., 2005). In other words, samples from a pen or a whole room are collected in order to increase sensitivity. An example of a technique like this is with the use of a pairs of gauze socks which swab the whole floor of a pen (Beloeil et al., 2004a). The disadvantage with this type of methods is that it is not targeting individual pigs, but groups of pigs living together. Moreover, the isolation of Salmonella in a room and generally in the environment does not always mean that there is an active infection of the pigs given the commonality of the pathogen in the environment (Griffith et al., 2006). This is even more challenging in the case of S. Typhimurium, where symptoms are usually missing and the pathogen is isolated in small numbers from the environment.
Another way to estimate the level of infection is the serological data. However, the collection of blood samples from live pigs would be difficult and would seriously affect their welfare. An alternative to this methodology is to check the serological status of a sample of pigs at slaughter. This is the principle on which the national monitoring programs of the EU member states are based on. However, this data is useful only as posterior information to characterise a farm and has limited value regarding the risk arising from individual pigs, given that by the time that antibodies are produced, pigs have stopped shedding the pathogen.